Understanding the Stakes of Medication Verification
Medication verification is the critical process of confirming that the correct drug, dose, and form are given to the right patient. It sounds simple enough until you consider the stakes. In the United States alone, medication errors cause approximately 1.5 million preventable adverse drug events annually. The financial toll is staggering, costing the healthcare system around $3.5 billion in excess costs every year. More importantly, research from the Agency for Healthcare Research and Quality (AHRQ) suggests these errors may contribute to up to 7,000 deaths in hospitals each year.
The goal isn't just to follow a checklist; it is to prevent harm. When we talk about verifying medication details, we aren't just talking about ensuring you have the bottle labeled "Acetaminophen." We are talking about ensuring it is the right strength, the correct dosage form, and free from look-alike confusion. As the Institute for Safe Medication Practices (ISMP) notes, the absence of clarity is often more dangerous than the wrong drug itself.
Breaking Down the Three Critical Elements
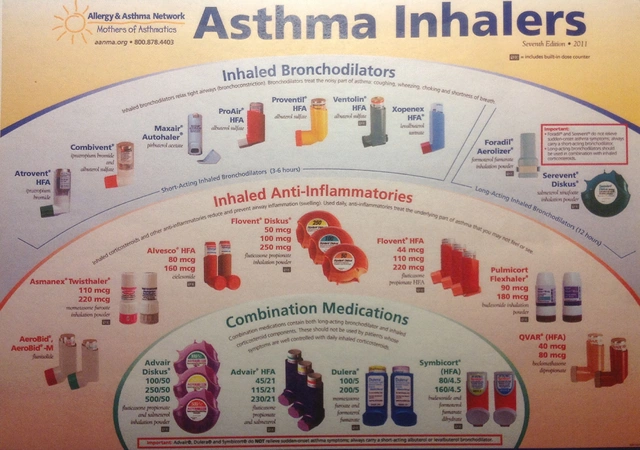
To verify a medication safely, you must dissect the order into three distinct components: name, strength, and form. Each carries its own risk profile.
1. Medication Names
Name confusion remains a leading cause of errors. A significant portion of mistakes happens because two drugs sound identical or look similar when written. These are known as Look-Alike Sound-Alike (LASA) drugs. For example, the difference between Cyanocobalamin and Methotrexate involves letters that can blur together in handwriting or small fonts. When checking names, never rely solely on your memory of what a prescription "should" say. Cross-reference the spelling against the original order.
Standardization helps here. Using systems like RxNorm normalization for drug names helps resolve discrepancies. One study found that combining standardized naming with spelling correction algorithms resolved over 92% of medication name issues. If you encounter an abbreviation that is ambiguous, such as "MS" which could mean Morphine Sulfate or Magnesium Sulfate, you must stop and clarify with the prescriber rather than guessing.
2. Strength Specifications
Strength verification is arguably the most math-heavy part of the process. You need to confirm that the amount of active ingredient per unit matches the doctor's intent. A common pitfall involves the ratio of concentration. For injectables, avoiding ratios like 1:1,000 in favor of explicit concentrations like 1 mg/mL is crucial. This change prevents errors involving epinephrine concentration, which accounted for hundreds of documented errors in recent years.
The format matters immensely. The FDA's guidelines explicitly require a space between the number and the unit (e.g., "10 mg" instead of "10mg"). This tiny whitespace prevents the eye from reading "10mg" as something else entirely. According to ISMP analysis of half a million error reports, this formatting rule alone prevents about 12% of misinterpretations.
3. Dosage Forms
The dosage form dictates how the medicine interacts with the body. An oral tablet cannot simply replace a transdermal patch even if they contain the same active ingredient. Confusion often arises when orders omit the form entirely. Research indicates that nearly 18% of reported errors involved missing dosage specifications, leading to scenarios where oral medications were administered intravenously or vice versa.
Always check the physical presentation. Is it a capsule, a scored tablet, or a liquid solution? The form affects absorption speed and bioavailability. Verifying this ensures the patient receives the therapeutic benefit intended.
A Practical Workflow for Safe Verification
You cannot verify medication effectively without a consistent routine. The American Society of Health-System Pharmacists (ASHP) mandates that verification occurs at three critical junctures.
- Receiving the Order: Before any action is taken, scan the request. Check for completeness. Does the order include the drug name, strength, dosage form, route, and frequency? If any of these fields are blank, flag them immediately. Missing information invites error.
- Preparing the Medication: When retrieving or compounding the drug, verify it against the electronic record or paper order. If you are using a barcode scanning system, ensure the scan matches before you even touch the vial. Hospitals using barcode medication administration (BCMA) systems saw an 83% reduction in dispensing errors compared to manual checks.
- Before Administration: Right before giving the drug, check again. Confirm the patient identity, the current stability, and that no new allergies have been added. A nurse shared a real-world story where cross-referencing the vial label ('Heparin 5,000 units/mL') against the order ('Heparin 50 units/mL') prevented a massive overdose.

Navigating High-Risk Traps and Shortcuts
Even experienced professionals slip up when shortcuts tempt them. Recognizing these traps is essential for maintaining safety.
Forbidden Abbreviations
Some abbreviations are banned for good reason. Using 'U' for unit is notorious; 'U' looks too much like the number zero or the letter 'O', leading to ten-fold dosing errors. Always write out 'unit'. Similarly, trailing zeros (like '5.0 mg') can be read as 50 mg if the decimal point is missed. Leading zeros (like '.5 mg') can be missed as '5 mg'. Always use '0.5 mg'. This simple formatting adherence addresses the fact that 34% of FDA-reported errors involve miscalculations of strength.
Tall Man Lettering
When dealing with high-risk drug pairs, look for Tall Man lettering. This typographic technique uses mixed case (capital and lowercase) to highlight the differences. For instance, writing predniSONE versus predniSOLONE. Data shows this method reduces confusion errors by 76%. Always pay attention when labels utilize this font style; it's a visual cue designed to slow your reading process and force recognition of the unique letters.
The Automation Bias Trap
We live in an age where computers help us sort drugs, but they aren't perfect. Dr. Robert Wachter, an expert in digital health, warns of 'automation bias.' This is when clinicians assume the computer is right and ignore visible red flags because the system approved the order. In 2020, The Joint Commission noted that alert fatigue contributes significantly to errors. Never let the green light override your own eyes. If a dose looks unusually high, pause and calculate manually.
Leveraging Technology While Maintaining Oversight
Technology is your biggest ally, but it requires human oversight. Electronic Health Record (EHR) systems integrated with clinical decision support tools can reduce medication errors by up to 55%. Epic Systems, for instance, cross-references thousands of drug names automatically. However, implementation varies.
Barcode scanning provides a hard check that the physical item matches the digital order. Yet, studies show that even with barcodes, incomplete data entry-specifically missing strength or form details-can bypass safety nets. In 87% of cases where drug name confusion led to errors, the strength information was omitted from the source prescription. Therefore, technology works best when combined with comprehensive training and mandatory fields in software that refuse to submit incomplete orders.

Fostering a Culture of Double-Check
Safety protocols are only effective when people embrace them. Implementing a four-eyes protocol, where two independent providers verify high-alert medications, has shown a 94% reduction in errors over an 18-month period in major medical centers. The most consistently praised technique remains the 'read-back' method.
In a 'read-back,' the person receiving the order verbally repeats the drug name, strength, and form to the sender. This auditory confirmation catches visual errors in spelling or decimal placement. It takes seconds but is cited as effective in nearly 90% of positive user experiences regarding error prevention. Create an environment where asking questions is encouraged. If a technician stops to ask about an ambiguous abbreviation, reward that caution rather than penalizing the time spent.
Frequently Asked Questions
Why is checking the dosage form as important as the drug name?
Dosage form determines how the body absorbs the medication. An oral tablet dissolves differently than an IV injection or a skin patch. Confusing these forms can lead to ineffective treatment or toxic overdoses because the absorption rate and concentration mechanisms differ significantly between forms.
What are the most dangerous abbreviations to avoid?
The most dangerous include 'U' for unit, which looks like a zero; 'µg' for microgram, which can be mistaken for 'mg'; and 'qod' or 'qd'. Always write out terms like 'every other day' and spell 'microgram' to prevent ten-fold dosing errors.
Can electronic systems fully replace manual verification?
No. While EHRs reduce errors by 55%, automation bias is a risk. Human verification is required to catch system glitches, typos in master lists, or scenarios where the system allows a technically valid but clinically inappropriate order.
How does Tall Man lettering help safety?
Tall Man lettering highlights differences in look-alike drug names using uppercase letters. For example, distinguishing enoxaPArin from enoxiParin. Studies show this method reduces confusion errors by 76% by forcing the eye to recognize the unique spelling sequence.
What is the 'read-back' method?
Read-back involves verbally repeating the medication order details (name, strength, form) to the prescriber upon receipt. This auditory check catches silent reading errors and misinterpretations, validating the communication loop between provider and dispenser.